FDA limits new vaccine reinforcements COVID to high -risk Americans only
NEWNow you can listen to News articles!
The United States Food and Medicines Administration (FDA) has authorized COVID-19 vaccines for autumn, but only for high-risk groups.
The Secretary of Health and Human Services, Robert F. Kennedy, announced the most recent FDA actions on Wednesday in an X publication.
“I promised 4 things,” Kennedy wrote. “1. to end COVID vaccine mandates; 2. Maintain the available vaccines for people who want them, especially vulnerable; 3. Demand the controlled tests with placebo of companies; 4. 4. To end the emergency.”
The largest measles outbreak in the United States is officially over, health officials say
“In a series of FDA actions today we achieved the four goals.”
In the publication, RFK declared that the FDA has issued “marketing authorization” for high -risk groups for the following vaccines: modern (6 months and more), Pfizer (5 years and more) and Novavax (12 and more).
“These vaccines are available to all patients who choose them after consulting with their doctors,” RFK wrote.
High risk groups include adults over 65 and those who are more likely to develop serious COVID diseases.
Medical Group goes against the CDC, recommends covid shots for young children
On their website, CDC lists conditions that can increase the risk of severe COVID, such as asthma, cancer, heart conditions, cerebrovascular disease, diabetes, dementia, mood disorders, obesity, parkinson and chronic diseases of the lungs, liver or kidneys, among many others.
RFK also announced that emergency use authorizations for COVID vaccines have been rescinded.
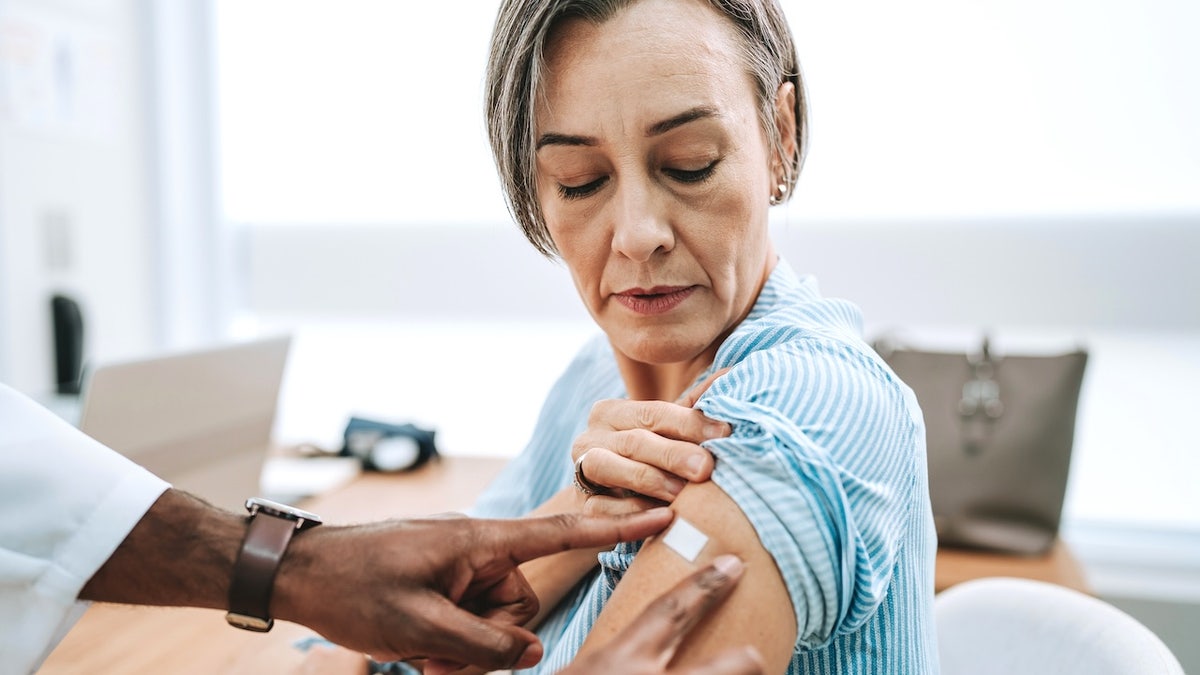
High risk groups include adults over 65 and those who are more likely to develop serious COVID diseases. (Istock)
“The American people demanded science, security and common sense,” RFK continued. “This framework offers all three.”
Before this change, the centers for disease control and prevention had recommended the vaccine for all Americans 6 months or more.
Click here to get the News application
In May 2025, Kennedy announced that COVID-19 vaccines would be eliminated from the CDC routine immunization schedule for healthy children and Pregnant women.
Click here to register in our health newsletter
Instead of a universal recommendation, the updated orientation of the CDCs requires “shared clinical decision making”, in which parents and doctors discuss the benefits and risks of vaccination for each individual case.
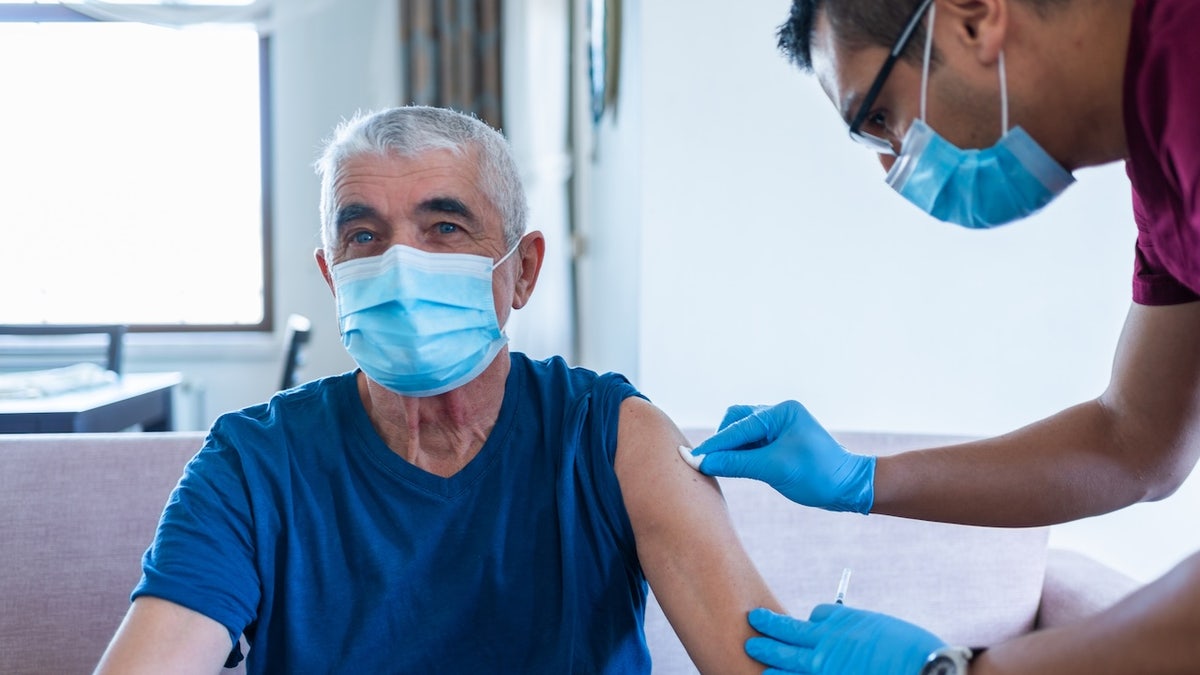
Before this change, the centers for disease control and prevention had recommended the vaccine for all Americans 6 months or more. (Istock)
However, the American Academy of Pediatrics (AAP) still includes it in its annual immunization program, as News Digital reported.
For more health articles, visit www.Newsnews.com/health
“It should be a conversation between the pediatrician, the patient and the father, and must depend on the health of the child and the current state of Covid,” said Dr. Marc Siegel, a senior medical analyst at News, News Digital.
Melissa Rudy is a senior health editor and a member of the lifestyle in News Digital. The advice of history can be sent to melissa.rudy@News.com.



