The CDC Committee moves to gradually eliminate the controversial mercury ingredient of flu vaccines
NEWNow you can listen to News articles!
A vaccine ingredient is at the attention center this week, since a preservative called Thimerosal was included on the agenda for a meeting of the Immunization Advisory Committee of the CDC (ACIP).
The “presentation on the thymusal in vaccines” was listed as an element of the agenda for Thursday, June 26, followed by “proposed recommendations on the influenza vaccine containing thimerosal.”
Timerosal (also thiomersal spelling) is a mercury -based preservative that has been used in multiple dose vaccines and medications since the 1930s as a means to prevent pollution, according to CDC.
CDC eliminates the recommendation of the COVID vaccine for healthy children and pregnant women
The ingredient is intended to prevent contamination by stopping the growth of bacteria and fungi, according to the same source.
Due to the growing concerns about the possible exposure to mercury, the American Pediatrics Academy, the Public Health Services Agencies and the vaccin manufacturers agreed that the thymatory should be “reduced or eliminated in vaccines.”
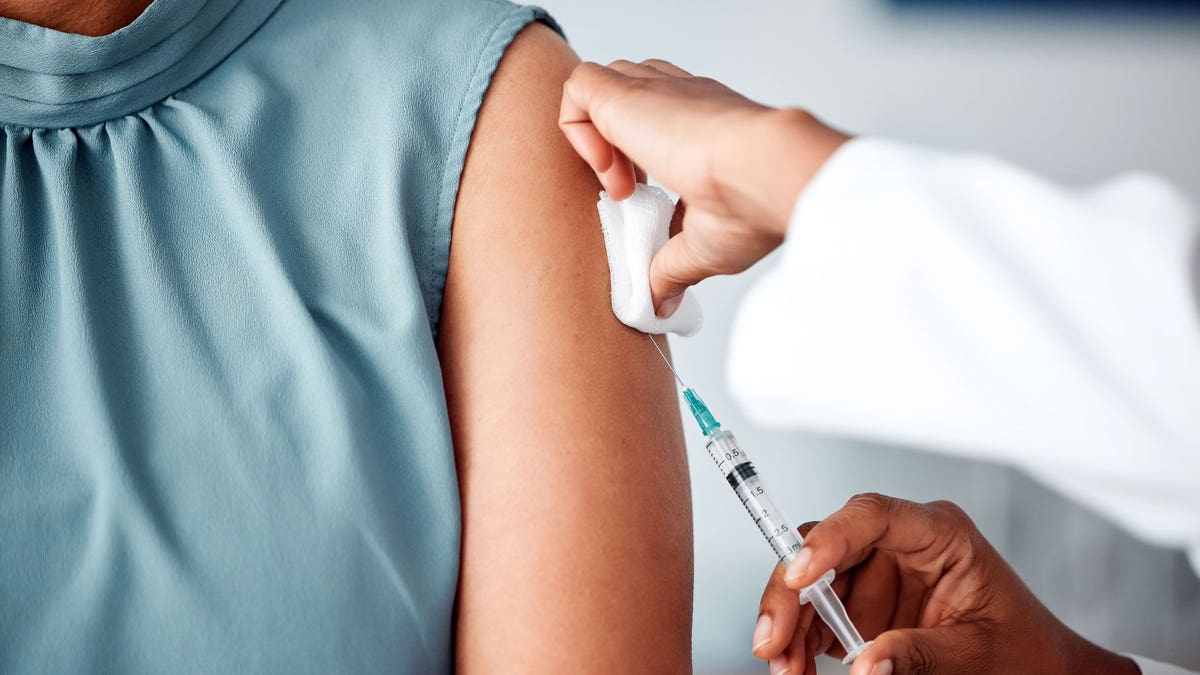
A vaccine ingredient is at the attention center this week, since a preservative called Thimerosal was included on the agenda for a meeting of the Immunization Advisory Committee of the CDC (ACIP). (Istock)
In 2001, the Timerosal was eliminated from all recommended vaccines for children 6 years or so, except for influenza.
Nowadays, multiple dose flu vaccines still contain timerosal, but other versions are available without the ingredient.
Click here to register in our health newsletter
At the June 27 meeting, the CDC Immunization Practices Advisory Committee voted to recommend that all adults (including pregnant women) and all children 18 years of age or less receive seasonal influenza vaccines only in single dose formulations that do not contain timerous, according to the notes of the meeting published by the CDC.
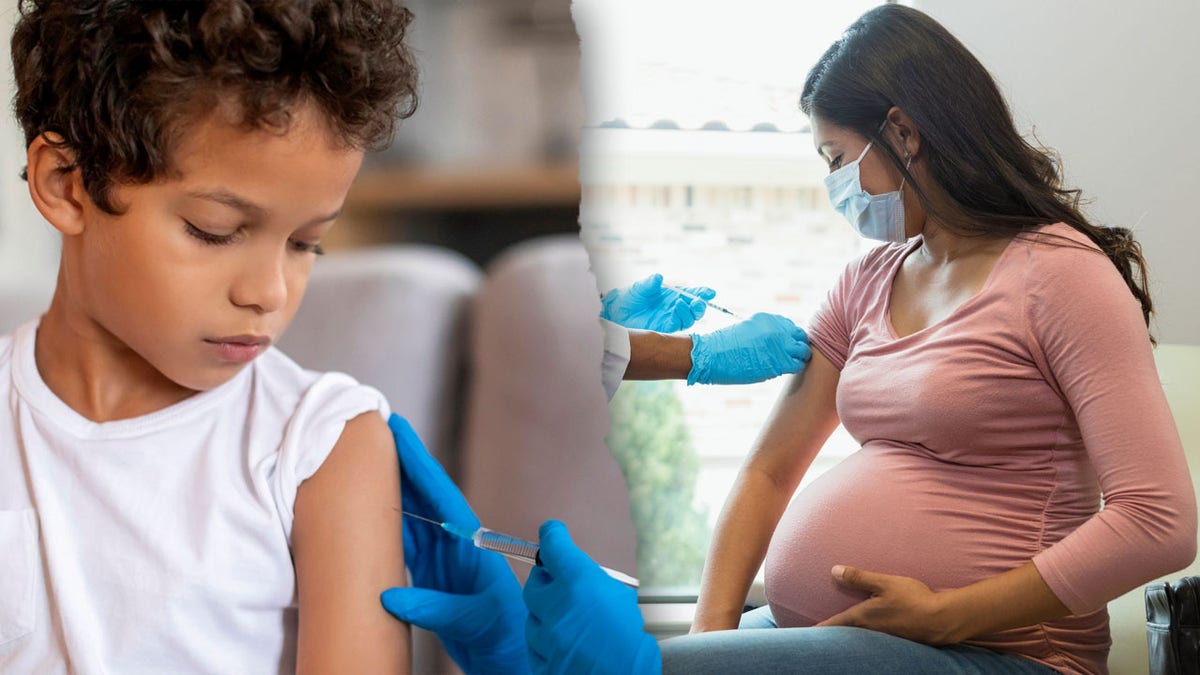
The Immunization Advisory Committee of the CDC voted to recommend that all adults (including pregnant women) and all children 18 years of age or less receive seasonal influenza vaccines only in single dose formulations that do not contain timerosal. (Istock)
Despite these recommendations, the CDC and other health agencies have affirmed that there is no evidence that the thimerosal raises health risks.
“The use of thimerosal in medical products has a record of being very safe,” says the agency’s website. “The data of many studies do not show evidence of damage caused by the low doses of thimerosal in vaccines.”
Click here to get the News application
Dr. Jacob Glanville, CEO of Centivax, a Biotechnology Company of San Francisco, reiterated that American children have not been receiving timerosal for more than 20 years in their vaccines, but remains in some multiple road vaccines, mainly outside the United States.

Nowadays, multiple dose flu vaccines still contain timerosal, but other versions are available without the ingredient. (Reuters)
“We support any effort to completely eliminate it: there are other preservatives that are not based on mercury,” Glanville told News Digital.
For more health articles, visit www.Newsnews.com/health
Glanville said that his company’s vaccines never contain mercury, aluminum, formaldehyde or materials derived from pigs or cows.
Melissa Rudy is a senior health editor and a member of the lifestyle in News Digital. The advice of history can be sent to melissa.rudy@News.com.



