The FDA approves the first injection twice a year that avoids HIV infection
FDA authorizes tool AI to predict the risk of breast cancer
Senior Medical Analyst Dr. Marc Siegel analyzes the advances in artificial intelligence aimed at predicting the future risk of breast cancer of an individual and the increase in cannabis health risks as users grow old.
NEWNow you can listen to News articles!
The United States Food and Medicines Administration (FDA) approved a new taking twice a year, the first and unique of its kind, to prevent HIV, the creator of the drug, Gilead Sciences, announced Wednesday.
Sold under the name of Yezugo, the company’s injectable inhibitor of the company’s HIV-1 capsid (Lenacapavir) reduces the risk of HIV sexually acquired in adults and adolescents.
“This is a historic day in the struggle of decades against HIV,” said Daniel O’Day, president and CEO of Gilead Sciences, based in California, in a press release.
Alzheimer’s disease could be avoided by the antiviral medication that is already in the market
The medicine, which should only be administered twice a year, has demonstrated “notable results in clinical studies”, since Gilead states that it could transform HIV prevention.
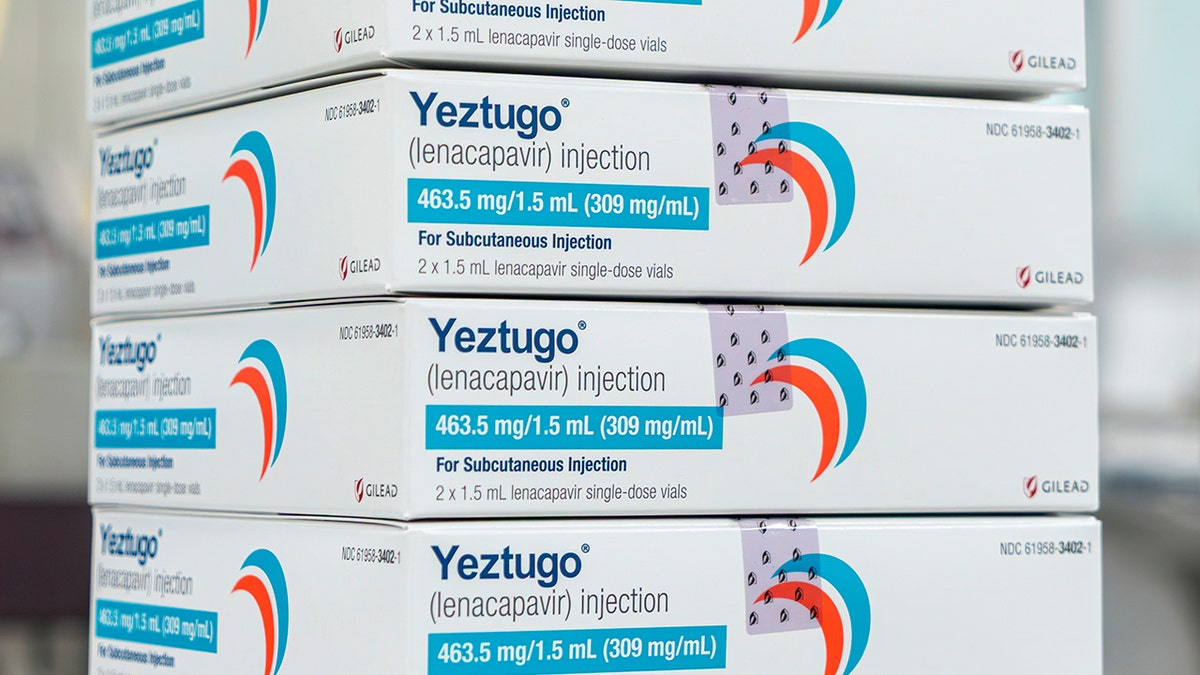
The United States Food and Medicines Administration has approved a new shot, twice a year, Yezugo, to prevent HIV, the creator of the drug announced on Wednesday. (Gilead Sciences through AP)
The medicine occurs as injectable under the skin that the body absorbs slowly. People must have a negative HIV-1 test before starting treatment.
Click here to get the News application
In large rehearsals last year, the medicine was not only almost 100% effective in HIV prevention, but also proved to be superior to oral medication once a day like Truvada, another Gilead medication.
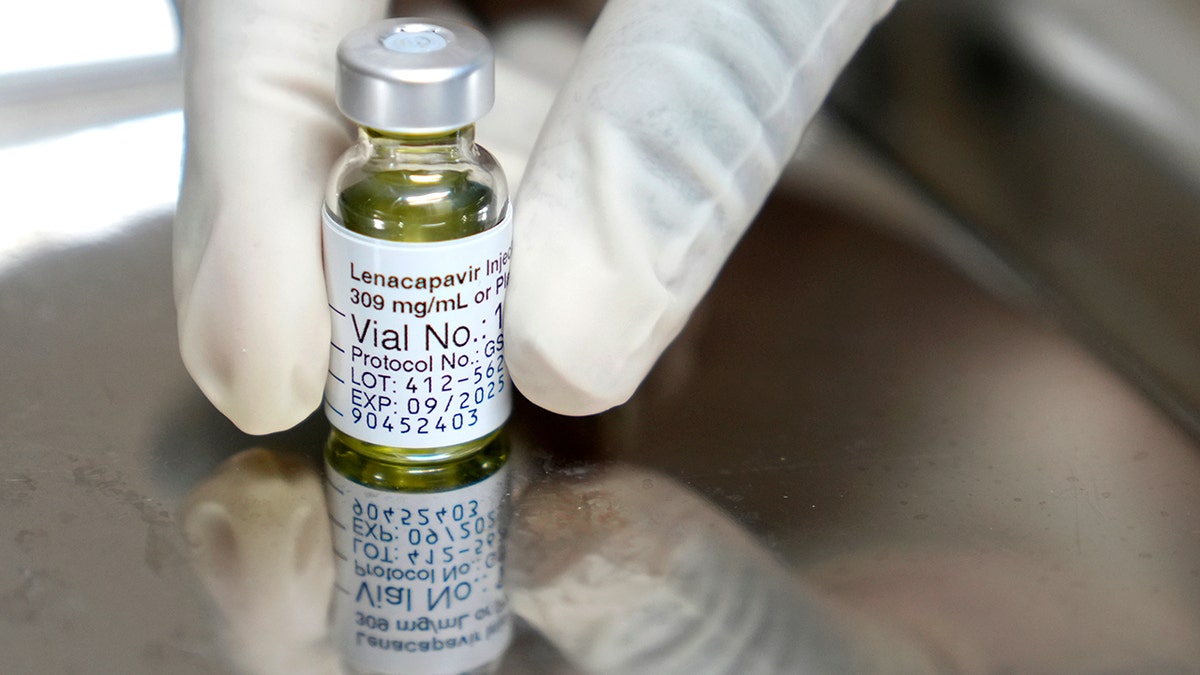
The medicine occurs as injectable under the skin that the body absorbs slowly. People must have a negative HIV-1 test before starting treatment. (AP Photo / Nardus Angelled)
Science magazine called Lenacapavir his “advance of the year” of 2024.
Click here to register in our health newsletter
Lenacapavir uses a several stages approach that distinguishes it from other approved antiviral medications.

Sold under the name of Yezugo, the company’s injectable inhibitor of the company’s HIV-1 capsid (Lenacapavir) reduces the risk of HIV sexually acquired in adults and adolescents. (Istock)
“While most antivirals act in a single stage of viral replication, Lenacapavir is designed to inhibit HIV in multiple stages of their life cycle,” says Gilead’s press release.
For more health articles, visit www.Newsnews.com/health
“Yezugo is one of the most important scientific advances in our time and offers a very real opportunity to help end the HIV epidemic,” O’Day said in the press release.
The most commonly reported adverse reactions during clinical trials included reactions of the injection site, headache and nausea, according to the company.
Khloe Quill is a lifestyle production assistant with News Digital. She and the lifestyle team cover a variety of stories issues that include food and drink, travel and health.



