The first HPV test in the home approved by the FDA could replace PAP test
Food and medication administration (FDA) has approved the first home detection test for cervical cancer, the product manufacturer announced on Friday.
The DIY test could serve as an alternative to the “Pap smear tests” in person, which are recommended every three years for women up to 65 years.
The self -length device, the blue green wand, made by Teal Health in San Francisco, allows women to acquire a sample and send it by mail for laboratory analysis.
The new cervical cancer treatment approach could reduce the risk of death by 40%, as shown in the test results
The test is designed to detect human papilloma (HPV), the virus that causes almost all cases of cervical cancer.
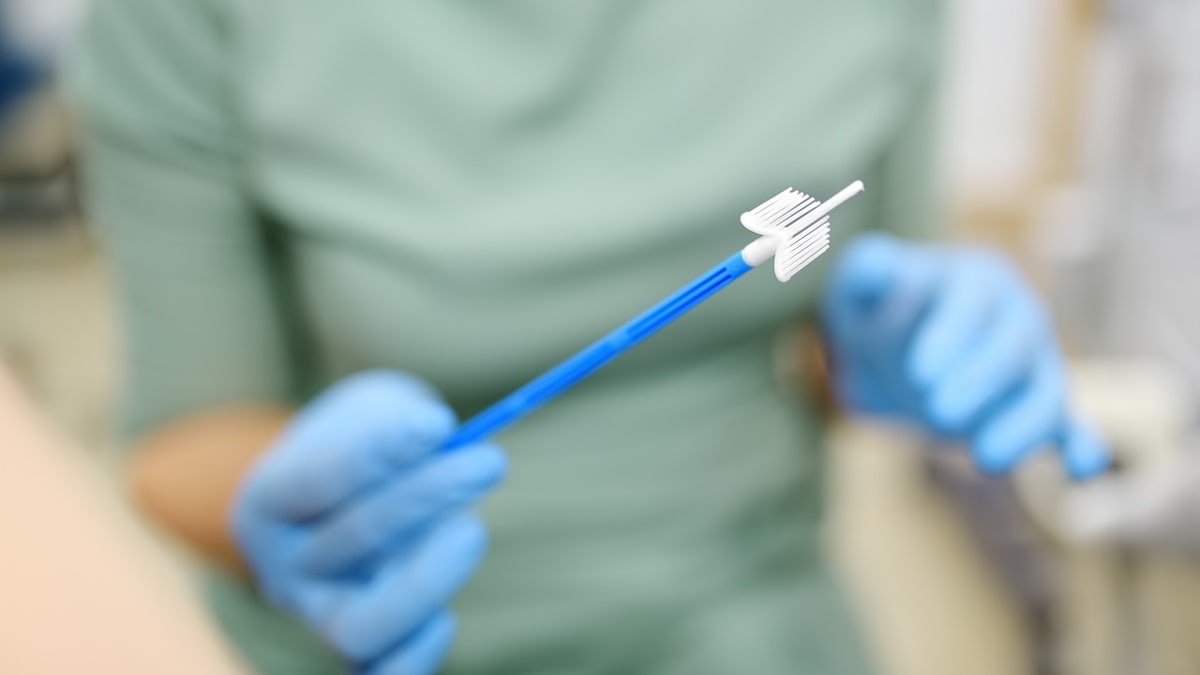
The food and medication administration approved the first home detection test for cervical cancer, the product manufacturer announced on Friday. (Istock)
The approval occurs after a clinical trial that includes more than 600 women, in which the blue green wand had a 96% detection of cervical precaence detection.
In the study, 86% of the participants said they would be more likely to meet the recommendations for the detection of cervical cancer if they could do it at home, Teal Health reported.
In addition, 94% said they would prefer to self -collection at home whenever the results were precise.
Cervical cancer deaths could be reduced with HPV tests at home, the study discovers
“Cervical cancer can greatly be prevented, however, detection rates in the US Colorado, which was a main investment in self -esteem in self -essay.
“It offers an evidence -based form of expanding access without compromising precision,” he added in a statement.

“Cervical cancer can be greatly prevented, but detection rates in the US. (Istock)
Approximately 11,500 new cervical cancer diagnoses are made in the US.
In addition to the risk of cervical cancer, some higher risk incidents of HPV can also cause other types of cancers, according to experts.
“Any type of test that helps detect cervical cancer is a victory.”
Women between the ages of 25 and 65 who have an average risk of cervical cancer will soon be able to order the test at home in www.getteal.comaccording to the announcement of the company.
The kits are expected to be available for the first time in California from June, with plans to expand throughout the country “as soon as possible,” said the company in its announcement.
Click here to register in our health newsletter
“Teal is working with the main insurance suppliers and plans to have flexible payment options, helping to eliminate financial concerns and ensure that more women have access to this preferred evaluation in the home if they wish,” said the company.

Approximately 11,500 new cervical cancer diagnoses are made in the US. (Istock)
In addition to the collection kit, the product also includes a Telesalud service with the support of medical suppliers throughout the process.
Women who give positive for HPV will be derived for a traditional papaser test. Those who do not positive are not considered at risk of cervical cancer and will not need to detect again for three or five years.
Click here to get the News application
“Some women are afraid of a traditional Pap smear or find that the process is uncomfortable; as a result, they postpone this vital test,” said Ami Vaidya MD, co -president of Gynecological Oncology at the John Theurer Center Medical Cancer Center of the Medical Center of the University of Hackensack, in a press release. (She was not involved in the trial).
For more health articles, visit www.Newsnews.com/health
“This could be an important tool to make more women regularly evaluate, especially those who do not have access to a medical provider. Any type of test that helps detect cervical cancer is a victory.”
Melissa Rudy is a senior health editor and a member of the lifestyle in News Digital. The advice of history can be sent to melissa.rudy@News.com.



